As we age, most of us think about wrinkles or sore joints, not the microscopic switches in our intestines. Yet a team of scientists in Germany and Italy has uncovered a core aging process in the gut that quietly turns off important genes in stem cells, reshaping the tissue from the inside out and raising the risk of colorectal cancer. The encouraging twist is that some of this “gut aging” looks like it can be slowed or even partly reversed.
The work is led by Prof. Francesco Neri of the University of Turin in Italy, together with researchers at the Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) in Jena, Germany, and the Molecular Biotechnology Centre in Turin. They analyzed intestinal tissue from humans and mice, combining DNA methylation maps, gene activity data, and organoid models to track how gut stem cells age over time.
Their findings, published in Nature Aging, describe a very specific pattern of epigenetic aging in the intestine that they call ACCA drift, short for Aging and Colon Cancer Associated drift. As Prof. Neri explains, “We observe an epigenetic pattern that becomes increasingly apparent with age.”
What It Means for the Gut to Age
The gut is one of the busiest tissues in the body. Every few days, specialized intestinal stem cells in tiny pockets called crypts generate a whole new layer of cells that line the intestines. On the surface, it looks like perfect renewal. But deep inside those stem cells, epigenetic marks on DNA slowly build up.
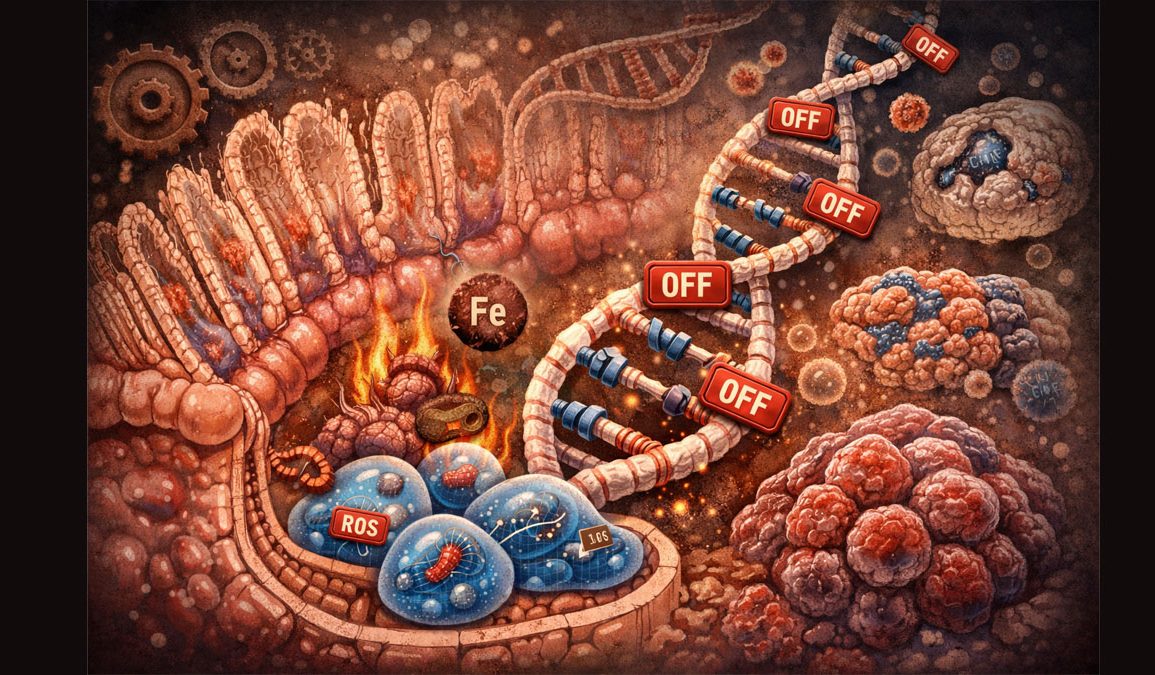
These marks are chemical tags that act like light switches, deciding which genes are turned on or off without changing the DNA sequence itself. Over time, the researchers found that a particular form of epigenetic drift slowly spreads in intestinal stem cells. This drift is dominated by DNA hypermethylation, where extra methyl groups pile up on gene promoters and shut them down.
In practical terms, an aging gut is not just “old tissue.” It is a mosaic of crypts where some stem cells still behave like those of a young person, while others carry an older epigenetic profile that limits self renewal and regeneration. The tissue becomes patchy and uneven in its ability to renew and repair itself.
Which Genes Fall Silent
The genes affected by ACCA drift are not random. They are heavily enriched in pathways that keep gut tissue healthy and balanced, especially the Wnt signaling pathway that is essential for stem cell activity.
Many of the drifted genes are involved in regulating how stem cells divide, differentiate, and maintain the intestinal lining. The study highlights entire gene families such as DKK and SFRP, which are known regulators of Wnt signaling. Their promoters become increasingly hypermethylated with age.
Across human colon samples, the team saw a consistent pattern. During normal aging, some promoters gained methylation and some lost it. But in colon cancer, the hypermethylated group exploded, and about 63 percent of promoters that were hypermethylated during aging were also hypermethylated in cancers. Nearly half of the ACCA drift genes were downregulated in cancer tissue.
This suggests that as these genes fall silent, they help create a cellular environment that is much more vulnerable to tumor development. Cancer samples looked epigenetically “older” than healthy tissue of the same chronological age.
A Patchwork Gut: How Aging Spreads Through Crypts
One striking observation is that ACCA drift does not affect the entire intestine evenly. Each crypt originates from a single stem cell. When that stem cell experiences epigenetic drift, the entire crypt inherits the new pattern. Over time, crypts divide and split, a process called crypt fission, and pass the drift along.
Dr. Anna Krepelova describes the result: “Over time, more and more areas with an older epigenetic profile develop in the tissue. Through the natural process of crypt division, these regions continuously enlarge and can continue to grow over many years.”
In older intestines, this produces a patchwork: some crypts remain epigenetically young, while others are heavily drifted and more likely to produce degenerated cells. The researchers showed that drifted crypts can gain a selective advantage and spread, forming clusters that behave much like crypts carrying oncogenic mutations.
How Iron and Inflammation Shut Down Repair
A key question is why these genes start to turn off in the first place. The team traced the problem back to iron metabolism and chronic, low level inflammation.
In aged intestinal cells, the researchers found that iron handling is distorted. Older cells absorb less iron from the environment but release more iron out of the cell. That leaves less bioavailable iron inside the nucleus, especially the ferrous iron (Fe²⁺) that acts as a cofactor for TET enzymes.
TET enzymes protect the genome from excessive DNA methylation by converting methylated cytosines into forms that can be removed. When iron levels drop, TET activity falls. As Dr. Krepelova puts it, “When there’s not enough iron in the cells, faulty markings remain on the DNA. And the cells lose their ability to remove these markings.”
This creates a domino effect. Reduced TET activity means methyl marks pile up and do not get cleared. Important genes for tissue maintenance and stem cell function are gradually locked in the off position. The researchers measured a significant reduction in TET enzyme activity in aged crypts, even though the enzymes themselves were still present.
Inflammation makes this worse. Mild inflammatory signals associated with aging, including interferon gamma, disrupt iron distribution in the cell and stress the metabolism. At the same time, Wnt signaling, which normally keeps stem cells active, grows weaker. This combination of iron deficiency, inflammation, and Wnt loss acts as an “accelerator” of epigenetic drift, causing gut aging to start earlier and spread faster than previously assumed.
Is Gut Aging Something We Can Mitigate
The study goes beyond description and shows that this gut specific aging pattern can be influenced. Using intestinal organoids, miniature guts grown from stem cells, the team tested ways to slow or reverse ACCA drift.
When they restored iron import by re expressing the transferrin receptor (TfR1) in aged organoids, nuclear iron levels rose. TET enzymes became more active again and the methylation marks on key gene promoters began to fall. Genes that had been silenced started to wake up.
Similarly, when the researchers boosted Wnt signaling using factors like Wnt3a or the small molecule CHIR99021, organoids from aged mice showed reduced promoter methylation and improved stem cell behavior. These interventions rebuilt iron homeostasis, revived TET activity, and pushed the cells back toward a younger epigenetic state.
Dr. Krepelova sums up the significance: “This means that epigenetic aging does not have to be a fixed, final state. For the first time, we are seeing that it is possible to tweak the parameters of aging that lie deep within the molecular core of the cell.”
Can These Silent Genes Be Switched Back On
Evidence from several angles suggests that the answer is yes, at least in controlled models. When TET enzymes were chemically inhibited in young organoids, promoter methylation on genes like Dkk2 and Sfrp1 increased in a dose and time dependent way, mimicking aging drift. Mice lacking TET2 and TET3 also showed higher methylation of these promoters.
On the other hand, when iron levels were restored or Wnt signaling was stimulated, TET activity rose and methylation on those promoters decreased. In aged organoids that regained transferrin receptor expression, the hypermethylated genes became more accessible again.
This shows that the silence of key genes is not simply a one way ticket. It is controlled by a balance between methylating enzymes and demethylating TET enzymes, which in turn depend on iron and signaling pathways. Shift that balance, and the epigenetic switches can move back toward an on position.
The authors see ACCA drift as one example of a broader principle. Epigenetic drift is a hallmark of aging across tissues, but this work argues that it is not just random noise. In the intestine, at least, drift is a structured, tissue specific program driven by concrete molecular events.
The team suggests that the body may carry multiple overlapping “subdrifts,” each with its own drivers and consequences. In the gut, the subdrift centers on iron homeostasis, TET activity, Wnt signaling, and inflammatory stress. Elsewhere, other mechanisms may dominate. Understanding this map of drifts could clarify why some tissues fail earlier than others and why certain cancers are tied so strongly to age.
Their data also hint that interventions targeting iron balance, inflammation, and Wnt signaling might one day help prevent or delay age related deterioration of the gut and its progression toward colorectal cancer. At the same time, they emphasize the limits of the current work. Many of the functional experiments were done in mice and organoids, and they note that more research is needed to confirm these mechanisms in humans and to show directly that ACCA drift drives cancer, not just accompanies it.
Still, the message is powerful. As our gut ages, essential genes in stem cells do not simply fade at random. They are silenced by a specific epigenetic program that we are beginning to understand and, in principle, to influence. Instead of seeing aging as a fixed fate written in our DNA, this work suggests it is at least partly a story of switches that, with the right tools, might be adjusted.