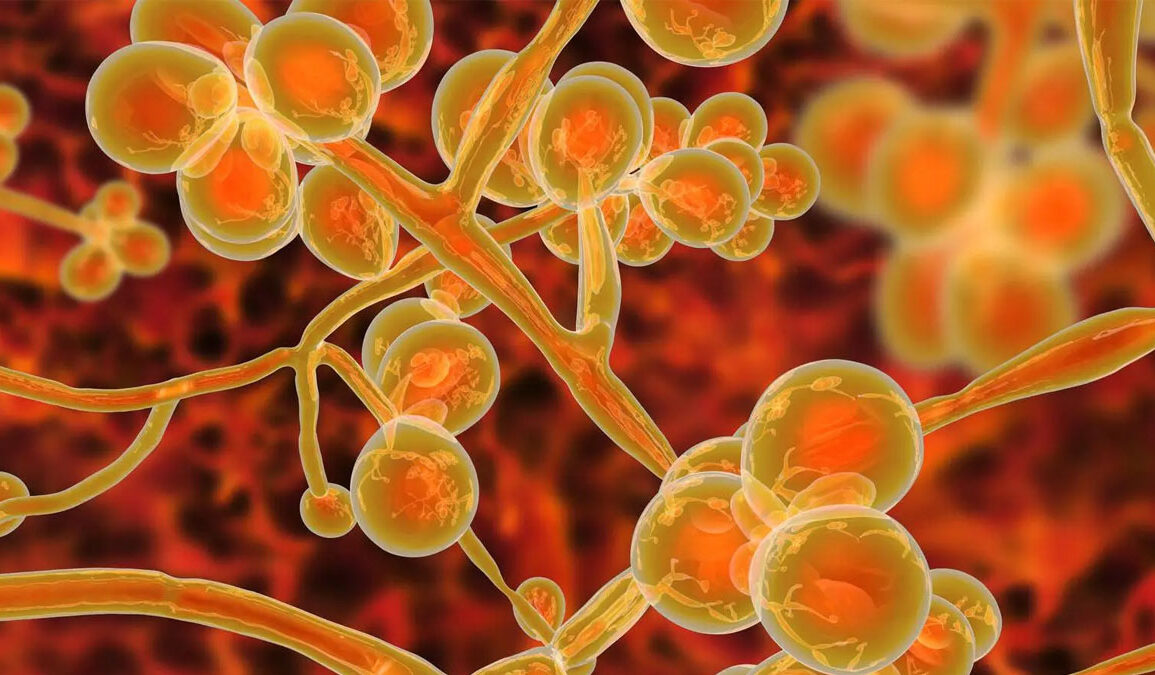
A growing body of research suggests that fungi might infect the brain, spark inflammation, and even help generate the toxic proteins seen in Alzheimer’s disease. Below is a clear tour of the key studies, who did them, what they found, which fungi were implicated, the strongest correlations and statistics, and what this could mean for future treatments and possible reversal of dementia symptoms.
Spanish team: fungal cells inside Alzheimer’s brains
Who: Diana Pisa, Ruth Alonso, Alberto Rábano, Izaskun Rodal, and Luis Carrasco
What they found: Using immunohistochemistry, confocal microscopy, and fungal-DNA sequencing, they detected fungal cells and hyphae both inside and outside neurons across multiple brain regions of Alzheimer’s patients. Fungal material appeared in the external frontal cortex, cerebellar hemisphere, entorhinal cortex and hippocampus, and choroid plexus. They also saw fungal elements in blood vessels. No fungal material was found in control brains.
Which fungi: Signals consistent with Candida glabrata, Candida albicans, Candida famata, Phoma betae, Syncephalastrum racemosum, Malassezia species, and Saccharomyces cerevisiae, among others. The authors emphasize mixed infections that vary by patient.
Correlations and statistics: In their hands, fungal cells or material were present in 100% of Alzheimer’s patients analyzed and absent in controls. They also note that up to 90% of Alzheimer’s patients show cerebrovascular pathologies, and they detected fungal structures within vessels, suggesting a possible link to vascular damage. They point to prior reports of chitin bodies in Alzheimer’s brains, elevated chitinase in blood and cerebrospinal fluid, and the antimicrobial activity of amyloid beta against Candida albicans.
Prospects for treatment: They cite two patients initially diagnosed with Alzheimer’s whose symptoms reversed after antifungal therapy, and argue that clinical trials with modern antifungal drugs are warranted because “there are a number of highly effective antifungal compounds with little toxicity.” They propose that chronic fungal infection could drive amyloid production as a defense, which later accumulates into plaques.
Baylor College of Medicine and collaborators: how Candida gets in and triggers Alzheimer’s-like changes
Who: Dr. David Corry and colleagues
What they found: In mouse models, Candida albicans can cross the blood–brain barrier by secreting enzymes called secreted aspartic proteases that degrade the barrier. Once inside, the fungus activates two microglial clearance pathways. The same fungal proteases also cleave amyloid precursor protein into amyloid beta–like fragments that stimulate microglia through Toll-like receptor 4, while a fungal toxin called candidalysin activates microglia through CD11b and is essential for clearing the fungus. In healthy mice, brain infection resolved in about 10 days, but the fungus generated amyloid-like peptides during the process.
Which fungus: Candida albicans
Correlations and statistics: This work explains a plausible route into the brain and a mechanism for producing amyloid-like peptides from an external source. It ties fungal entry and persistence to the same molecular actors long associated with Alzheimer’s pathology.
Prospects for treatment: The team suggests that enhancing these microglial clearance pathways or preventing Candida’s barrier breach could be therapeutic. They propose that amyloid may be generated from both brain enzymes and fungal proteases, which opens new targets for intervention.
Alzheimer’s Pathobiome Initiative: documented reversals after anti-infective therapy
Who: The Alzheimer’s Pathobiome Initiative, led by Nikki Schultek with clinicians and scientists from institutions such as Massachusetts General Hospital, Harvard Medical School, and Oxford
What they found: By compiling cases in which dementia-like symptoms were linked to brain infections, they report that patients often improved after antiviral or antifungal treatment. University of Edinburgh molecular biologist Richard Lathe noted that in several cases symptoms “went away” following anti-infective therapy.
Which microbes: The group highlights both fungi and bacteria; Candida albicans has been repeatedly implicated in related literature.
Correlations and statistics: Because this challenges the dominant view of Alzheimer’s, exact percentages are not yet known. Lathe’s view is that “it’s unlikely to be 100 percent,” but he ventured that “probably half or more could potentially be treated,” stressing that further research is needed.
Prospects for treatment: If confirmed, routine screening for brain infections and trials of targeted anti-infective regimens could make certain dementias reversible in a subset of patients.
Additional evidence that supports or challenges the hypothesis
Detection of fungal sequences: Independent work using rDNA locus amplification and deep sequencing has reported fungal sequences in human brain tissue, consistent with a brain mycobiome signal.
Microbes and immunity: Reviews on neuroimmune contributions to Alzheimer’s emphasize elevated cytokines and chronic inflammation in patients, which aligns with the idea that persistent infection could be a driver for ongoing immune activation.
Animal and ecological clues: Mouse studies show Candida albicans can enter the brain when immunity is compromised. A preprint described higher loads of bacteria, viruses, and fungi in brains from Alzheimer’s patients than in healthy controls. Other experiments in fish demonstrate that microbes can take up long-term residence in brains without obvious early consequences, making silent accumulation plausible with age.
Skepticism and alternative explanations: Some microbiologists caution that microbial fragments are ubiquitous and contamination must be ruled out. Others suggest that damaged barriers or weaker immunity in Alzheimer’s could allow more microbes in without microbes being the root cause. The debate underscores the need for rigorous, well-controlled human trials.
Numbers at a glance
- Fungal material in Alzheimer’s brains across regions and inside neurons: detected in 100% of patients analyzed by the Spanish group, absent in controls.
- Vascular pathology in Alzheimer’s: up to 90% of patients show cerebrovascular lesions, and fungal elements were found in vessel walls in Alzheimer’s tissue.
- Mouse infections by Candida albicans: brain infection cleared in about 10 days in healthy mice, while generating amyloid beta–like peptides during the response.
- Reported human reversals: case reports and compiled cases describe dementia symptoms improving after antifungal or antiviral therapy, motivating formal trials.
What fungi are most often implicated?
- Candida species feature prominently, especially Candida albicans and Candida glabrata.
- Other reported or detected genera include Candida famata, Malassezia spp., Phoma betae, Syncephalastrum racemosum, and Saccharomyces cerevisiae. Findings vary by patient, suggesting mixed infections are common.
How could this change care?
Screening and diagnosis: If a fungal contribution is validated, clinicians could screen for fungal DNA, proteins, or polysaccharide markers in blood, cerebrospinal fluid, or possibly through imaging-guided biopsy in select cases. Elevated chitinase, prior reports of chitin bodies, and circulating fungal macromolecules could serve as clues.
Therapeutic trials: Modern antifungals with favorable safety profiles could be tested in randomized, controlled studies for patients with biomarker evidence of fungal involvement. The Baylor work points to additional targets, such as blocking fungal proteases that degrade the blood–brain barrier or modulating microglial receptors that control clearance.
Prevention and risk reduction: Strengthening overall immune defenses with vaccines or lifestyle strategies may help reduce late-life brain colonization. Managing other modifiable risk factors remains important, since sleep, metabolic health, blood pressure, and social engagement all affect dementia risk.
Bottom line
The fungal hypothesis does not claim that every case of Alzheimer’s is infectious. It argues that in at least a subset of patients, brain fungi may seed chronic inflammation, damage vessels, and even help generate amyloid-like peptides. Human brains from Alzheimer’s patients have repeatedly shown fungal material that is absent in controls, animal work maps out a plausible mechanism, and documented patient reversals after anti-infective therapy suggest real clinical stakes. The next decisive step is carefully designed clinical trials that pair fungal biomarkers with targeted antifungal treatment and track cognition. If successful, this line of research could unlock new treatments and, for some patients, true reversal of dementia symptoms.